TOC Purified Water analysis as it should be, according to USP and EP
TOC is one of the most important quality parameters when it comes to Purified Water (PW) and Water for Injection (WFI). Internationally harmonised standards demand fast and accurate online analysis of water flow. Constant developments in the digitisation of (production) processes are also being embraced by the various pharmacopoeias, hence the buzzword "PHARMA 4.0" by analogy with INDUSTRY 4.0. Data integrity is a main concern in this regard.
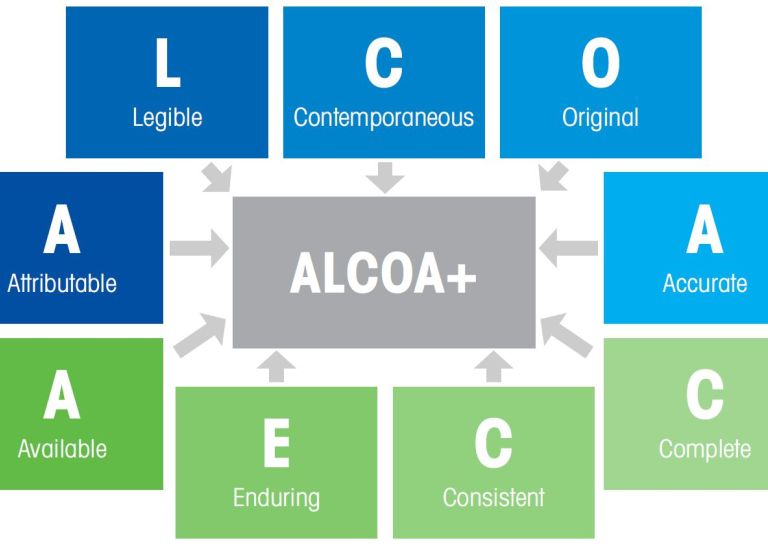
ALCOA+
The basis of the legal framework is the well-known CFR21 part 11. ALCOA and ALCOA+ are the framework within which these regulations are to be developed. Pharmaceutical manufacturing companies are further converting this into their own SOPs (Standard Operating Procedure).
| ALCOA | ALCOA + |
|---|---|
| Attributable | Attributable |
| Legible | Legible |
| Contemporaneous | Contemporaneous |
| Original | Original |
| Accurate | Accurate |
| Complete | |
| Consistent | |
| Enduring | |
| Available |
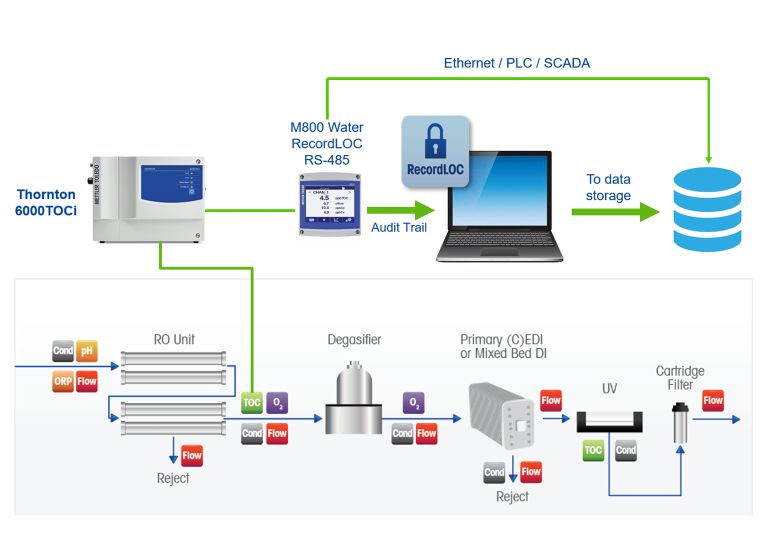
RecordLoc
Pharmaceutical companies have invested a great deal of money to align their process control systems and data acquisition with international regulations such as CFR 21 part 11. They would obviously want to continue to use those systems. And rightly so! That is why TOC analysers guarantee data integrity through a connection to an (industrial) PC, with an Audit Trail app such as RecordLOC. A not insignificant plus point here is that through the same system, the conductivity of ozone (O3) and TOC analyser can be connected simultaneously and in this way all the most important analyses are and remain ALCOA+ compliant.
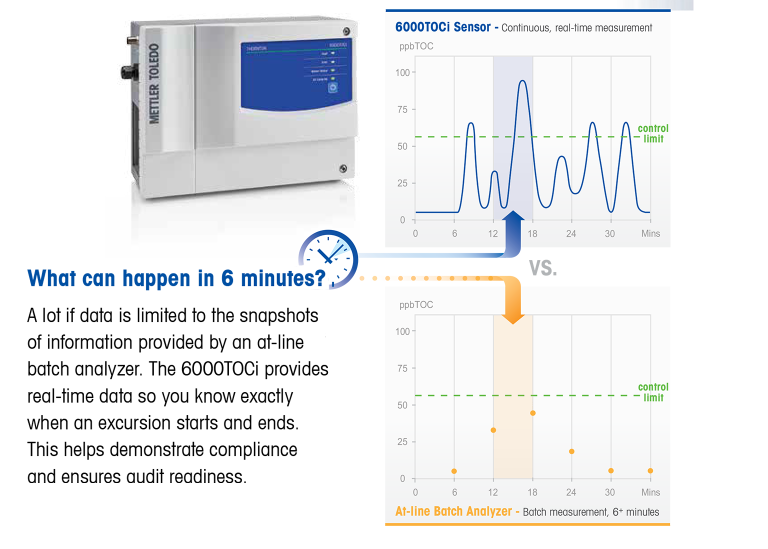
The TOC analyser
With a PW or WFI water flow, you want to know the TOC at any time, without delay. It simply will not do to take a sample every six minutes and then analyse it, even though this is done fully automatically. For such an important quality measurement, you need an actual online analyser that measures the water as it flows through it. After all, consider all that could happen between samplings.
Furthermore, it is important for a quick response time that the analysis is done as close to the water flow as possible. The control panel and data processing can easily be placed a little further away, e.g. in a technical room. It only becomes really interesting if this allows you to link other sensors such as conductivity or ozone (O3) to the same system.
SST
It goes without saying that you must be able to rely 100% on the analysis results. Regular calibrations and SSTs (System Suitability Tests) are therefore essential. The USP, EP and JP (the so-called "tripartite") all accept sucrose and p-benzoquinone as SST standards because they are non-conductive. More conductive reagents, e.g. KHP (potassium hydrogen phthalate), are not suitable for all types of analysers. EP and USP do not want to exclude any technology, which is why sucrose and p-benzoquinone are better suited for SST than KHP.
Reduced water consumption
Water, especially if it is of the purest kind, is expensive. As a manufacturer of pharmaceuticals, you try to work sparingly with it, because every litre counts. It may seem like a minor detail, but the water consumption of a TOC analyser is actually a determining factor when choosing one. Companies around the world are increasingly opting for analysis systems that consume less than 10ml/min, thereby requiring up to 7 times less water than competing systems.
Conclusion
Are you commissioning a new water system? Or is the current setup in need of an upgrade? Or are you looking to comply with increasingly stringent regulations? Then it is best to opt for a "future-proof" TOC analyser. Fast response time, ALCOA+ / CFR21 part 11 ready and carbon footprint are certainly criteria that are considered and strongly influence the choice. The Mettler Toledo-Thornton TOC6000i analyser, available in Benelux through ELSCOLAB, meets all requirements for a modern PW or WFI system.
More information?